Not all bugs are bad: Development, the gut-brain axis and mental health
Ben Hollingdale talks us through the relatively new field of gut microbiome science and explores how gut flora can affect both our physical and mental health.
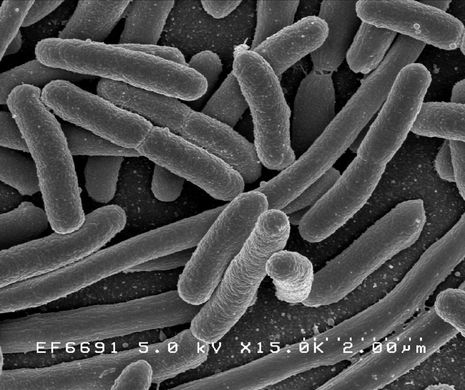
While the devastation caused by the novel coronavirus (SARS-CoV-2) continues to be felt by our social and political institutions, ongoing research into the science of gut microbiomes paints a rather different picture of human-microbe interaction.
Over 90% of the cells in the human body belong to micro-organisms. The majority of these microorganisms – bacteria, viruses, fungi and even microscopic animals (helminths and flatworms) – constitute our normal flora, a micro-environment of mostly benign (or “commensal”) organisms that we carry with us at all times. It is important to note that this “commensalism” is a largely location-specific phenomenon. You may have read, for example, that around 10% of adults carry meningococcus (Neissaria meningiditis) – the bacteria responsible for meningitis – inside their nasal cavity and on the surface of their skin at all times. However, what is regarded as normal flora for the nasal cavity or the gastrointestinal tract can cause serious life-threatening infection if it enters a reasonably sterile environment, like blood or cerebrospinal fluid.
The majority of commensals in the human body live in and are associated with the mucous membranes of the gastrointestinal tract (or simply the gut). These mucosae are colonised by exposure to micro-organisms in the early stages of life. While bacteria present in the prenatal meconium, umbilical cord blood, and amniotic fluid suggest that the microbial succession of the gut may start prenatally, the majority occurs during and after birth, either with the vaginal microbiota (in traditional delivery) or the skin flora (in the case of C-section) of the mother; postnatally it occurs with micro-organisms in colostrum and breastmilk, and later in solid foods.
“The question remains: what exactly explains the link between an unhappy gut and an unhappy mind?”
The colostrum is rich in proteins, including antibody and complement, which prevent the excessive growth of microbes while the immune system of the neonate is not fully developed. The high numbers of Bifidobacterium acquired from the vaginal microbiota during birth also prevent the growth of pathogenic gram-negative bacteria through competitive exclusion. Later on, bacterial growth is aided by carbohydrates present in breastmilk – over 200 human milk oligosaccharides (HMOs) – which pass through the gastrointestinal tract mostly undigested before being broken down by extracellular proteolytic enzymes produced by microorganisms in the colon. These carbohydrates fuel the growth of key gut bacteria and metabolites, such as free fatty acids (FFA), which facilitate the rapid development of the brain, gut, and other key structures in the growing child. As the infant transitions onto solid foods, the proportion of Bifidobacterium in the gut flora decreases, and the microbiome becomes more diverse and complex. The adult microbiome is fully developed around 2-3 years after birth.
Dysbiosis, a disruption of the stable micro-environment of the gut, has been considered a culprit in the aetiology of a number of illnesses since the gut microbiome was first discovered in the mid-19th Century. The adult gut is mostly dominated by bacteria belonging to two groups: Firmicutes and Bacteroidetes. These share a common evolutionary ancestor. The variable composition of these groups within the gut (particularly Prevotella, a genus of Bacteroidetes) between individuals has been linked to different diets across the world as well as early-life adversity.
In recent years, patent advances in the “omics” technologies have enabled greater characterisation of the composition of gut flora both in health and illness. The publication of the Human Microbiome Project in 2012 fuelled a new wave of interest in the field of gut microbiome science and the role of “good” and “bad” bacteria in a genesis of conditions, ranging from liver-disease and periodontis to obesity, inflammatory bowel disease and cancer. Some opportunists have cashed in on the link with obesity specifically, offering to use faecal examinations to help with weight loss – you may recall this from the since discontinued television program You Are What You Eat, featuring Gillian McKeith. However, with a lack of clinical studies on humans, this has yet to be implemented in widespread medical practice.
“Gut health might not be the ‘golden ticket’ to a happier mind, but it does paint a slightly more heart-warming picture of the ways humans interact with microbes.”
In popular science, much of this excitement has been concentrated around the ability of gut microbe composition to affect mental health. Experiments with faecal microbiota transfer (FMT) in mice have shown that transferring gut microbiota from the faeces of a healthy individual to the gut of a dysregulated individual can mitigate the development of a range of psychiatric disorders (including depression, schizophrenia, and anorexia nervosa). A lot has been made of this connection. Potential psychobiotic therapies, including using FMT in humans, promise to treat these conditions by treating the gut microbes first, but as Ted Dinan of University College Cork points out, most of these are still in the preclinical stage. With the wide-ranging effects of gut microbe composition on human health, there are risks of using these therapies to treat mental illness when the exact pathways involved are yet to be elucidated. Hence, it will likely be some time before we see psychobiotic therapy being used in clinical practice.
That being said, the complexity and diversity of the gut microbiome has been convincingly linked to a range of psychiatric conditions in humans. Trauma to the gut microbiota during development is linked to an increased incidence of these conditions in later life. A 2019 nationwide study in Denmark showed that individuals who were hospitalised for bacterial infections in childhood were at increased risk of developing mental illnesses as their gut flora was degraded by strong courses of antibiotics. The question remains: what exactly explains the link between an unhappy gut and an unhappy mind?
Recent research exploring the reasons behind the link points to the influence of a “gut-brain-microbe axis.” While all of the pathways involved are yet to be characterised in full, this gut-brain-microbe axis describes a system of bidirectional communication between the emotional and cognitive centres of the brain and the microbes inside the gut. These communications are modulated by the central nervous system (CNS), which includes the brain and the spinal cord, and the enteric nervous system (ENS), which has been shown to control gastrointestinal functioning independently of CNS control.
The microbes in the gut may interact with the ENS through the production of metabolites which modulate ion channel activity or act as local neurotransmitters and these are communicated to the brain through connections of the ENS and CNS. The brain responds primarily with the release of signalling molecules, which, through a range of actions, alter the local environment of the gut microbes. If the composition of microbes in the gut is offset, the communications received by the brain are interpreted as markers of physiological stress. Likewise, in chronic stress, communications in the other direction can create an unfavourable environment for the gut microbiota.
The field of gut microbiome science is rapidly growing, but as a comparatively new discipline, there are some teething problems left to overcome before its use becomes widespread. Gut health might not be the “golden ticket” to a happier mind, but it does paint a slightly more heart-warming picture of the ways humans interact with microbes. After all, we simply couldn’t live without them!
 News / Union elections underway with only one position contested14 March 2026
News / Union elections underway with only one position contested14 March 2026 Lifestyle / Blind Date: ‘In an ideal world there would’ve been a little flirting’15 March 2026
Lifestyle / Blind Date: ‘In an ideal world there would’ve been a little flirting’15 March 2026 News / Uni urged to help locate looted Zimbabwean skulls14 March 2026
News / Uni urged to help locate looted Zimbabwean skulls14 March 2026 News / ‘Race realist’ fellow: College ‘actively encouraged’ protests for my sacking30 January 2026
News / ‘Race realist’ fellow: College ‘actively encouraged’ protests for my sacking30 January 2026 News / Union debates officer resigns after misconduct investigation9 March 2026
News / Union debates officer resigns after misconduct investigation9 March 2026